The new clinical research day hospitalization unit
Since May 2, 2022, a new unit has been welcoming clinical research patients on the2nd floor of the Center. This is the Investigation, Research, Innovation and Care Unit, known as the "IRIS Unit".
Patients enrolled in therapeutic trials are now cared for in this same unit. The IRIS unit has :
- its own sampling center,
- medical and paramedical consulting rooms,
- 10 day hospital beds/places,
- with a dedicated team of professionals with expertise in clinical research.
How are patients cared for in the IRIS Unit?
First of all, the patient's oncologist must present and explain the study to the patient, who then signs the consent form if he or she so wishes. The patient is then included in the study. Then :
- The patient is welcomed by the nursing team at the sampling center. Before each medical consultation, vital parameters (pulse, blood pressure, temperature) are measured, and examinations (ECG electrocardiograms, for example) are carried out.
- The treatment is administered under close supervision (several times an hour) by the nursing and medical teams (vital parameter measurements, patient feedback, blood sampling, etc.).
The follow-up of patients included in therapeutic trials can last several years, in order to assess the efficacy of treatments and measure any medium- or long-term toxicity. All patient-related clinical research activities are carried out in a single department dedicated to clinical research.
The benefits of IRIS
This new department, in an environment conducive to the safe care of patients, enables :
- improve conditions for patients and staff,
- improve the flow of care for clinical research patients.
" I'm delighted to see this IRIS unit open, optimizing access to the most innovative cancer treatments and providing optimum conditions for our patients," explains Dr Mélanie DOS SANTOS, head of the IRIS hospitalization unit.
Inauguration of the IRIS unit on May 13, 2022
The unit was inaugurated on Friday, May 13, in the presence of members of the Center's Board of Directors, the press and outside personalities. The inauguration began in the boardroom, with speeches by Dr Carine SEGURA-DJEZZAR, President of the CME, Pr Florence JOLY, Head of the Teaching, Research and Innovation Department, and Pr MAHE, Director General.
Organization of clinical research at the Centre Baclesse
The Clinical Research Department at the Centre François Baclesse is under the responsibility of Pr Florence JOLY, Head of the Teaching, Research and Innovation Department. Its members are :
- an investigation unit (IRIS Unit)
- and a promotional unit.
The IRIS unit
The care of patients included in type 1 interventional research on drugs or innovative therapies is provided by a dedicated medical and paramedical team:
This organization enables us to provide care for research patients from Monday to Friday, 8.30am to 7pm. Outside these hours, as well as on weekends and public holidays, continuity of care is ensured by the medical and paramedical teams of the departments involved in the patient's initial care, the on-call resident on site, and a senior doctor on call in accordance with the establishment's procedures.
Background to the opening of the IRIS Unit
The Centre François Baclesse has been a major player in clinical cancer research for over twenty years.
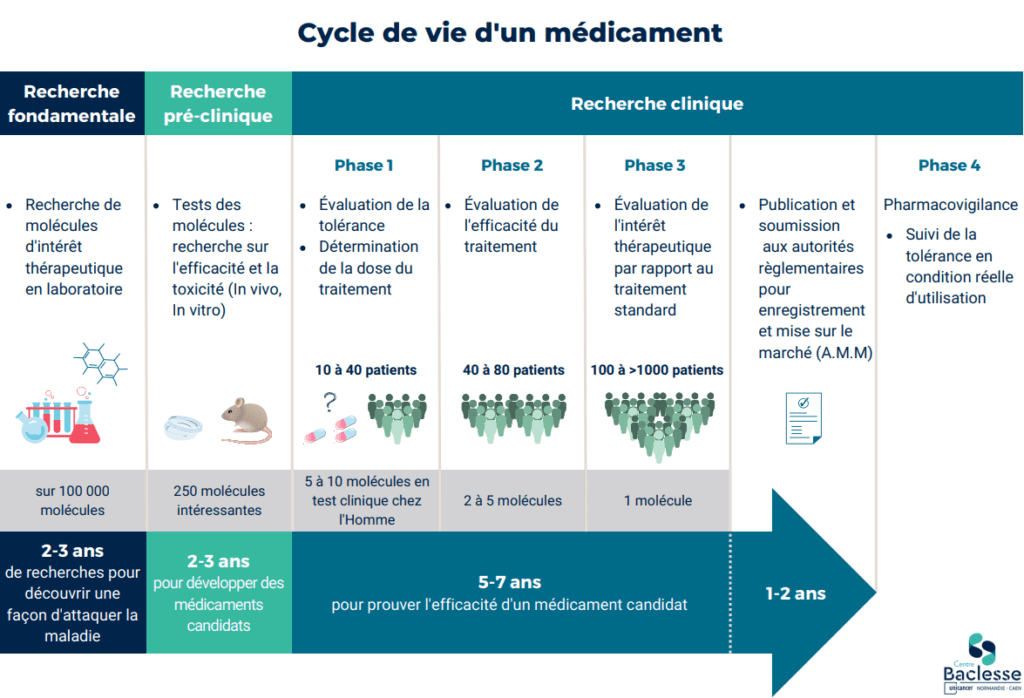
The clinical research department at the Centre Baclesse handles all categories of interventional studies, from phase I to phase IV, with priority given to early-phase trials. Every year, 170 interventional trials are open for inclusion, giving patients in the region access to innovative treatments in a wide range of therapeutic indications.
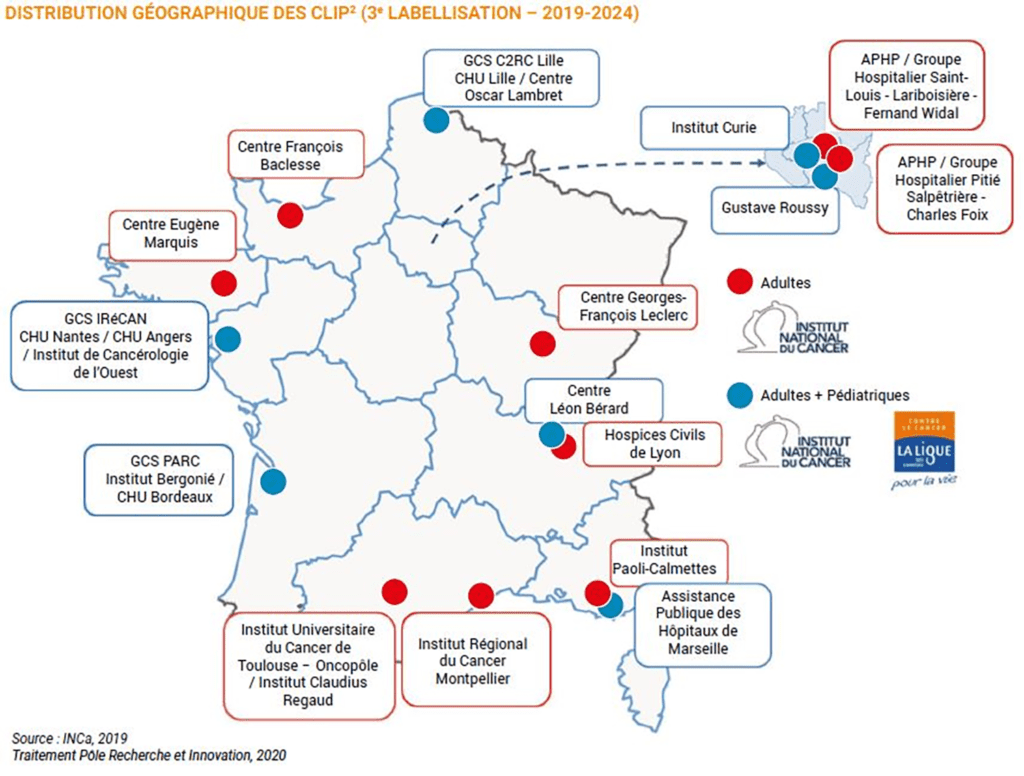
Since 2010, and for the3rd time in a row, the Centre Baclesse has been awarded the CLIP² label, "Labellisé de Phases Précoces en cancérologie adulte", by INCa, the French National Cancer Institute. The Centre Baclesse is the only one in Normandy to receive this label. This label distinguishes the 16 best French centers capable of conducting early-phase clinical trials in oncology, and thus offering their patients access to therapeutic innovation. The aim of early-phase trials is to assess the safety of a new drug (administered alone or in combination) and to obtain data on its anti-tumor activity. These trials are usually offered to patients when they represent an interesting alternative in their treatment, or when conventional treatments have failed. The previous label enabled the inclusion of over 400 patients in more than 80 early-phase trials over 4 years.
Note that since 2017, clinical research at the Centre Baclesse has been ISO 9001:2015 certified by AFNOR. This internationally recognized standard guarantees everyone - patients, doctors, academic and industrial partners - a high level of quality and safety in the organization of clinical trials.
At the Centre François Baclesse, patients involved in interventional research on health products are cared for by a dedicated medical and paramedical team, supplemented by staff from the Centre François Baclesse. Until now, this activity was integrated into the day hospital department, located on the first floor of the establishment, with a surface area of 80m² and 6 dedicated beds.
From May 2, 2022, the entire clinical research team will be housed on the 2nd floor of the facility, with a surface area of 310 m² and 10 outpatient hospitalization places, enabling us to increase our capacity in line with the development of our activity and improve our organization in terms of patient care and monitoring.
" This is the culmination of a process initiated by the establishment over 10 years ago to integrate therapeutic innovation into patient care. This new department will enable us to pursue our ambitions by giving more patients access to clinical research," says Prof. Florence JOLY, Head of the Teaching, Research and Innovation Department at the Centre Baclesse.
Why develop clinical research at the Centre Baclesse?
Research is one of the three missions of cancer centers, alongside care and teaching.
Patient participation in clinical trials is an essential contribution to the development of new treatments and therapeutic strategies. These innovative therapies will benefit a large number of people affected by cancer. Early-phase trials give patients access to innovative treatments not available on the market. Advances in cancer treatments are the fruit of clinical research, thanks to trials conducted with rigor and method, guaranteeing a high level of scientific proof. Today's treatments are yesterday's clinical trials.
Clinical research objectives :
- Advancing medical knowledge
- Offer patients access to innovative treatments well before they reach the market (more effective, better tolerated).
- Determining the best therapeutic strategy (establishing guidelines)
In therapeutic trials, treatments may be :
- New molecules,
- Drug combinations,
- New ways of administering them or new treatment techniques (surgery, radiotherapy, etc.).