One year after the opening of the TRIPLEX trial, 37 patients with triple-negative breast cancer are already included in this research protocol with organoids...
A look back at the study and triple-negative breast cancer
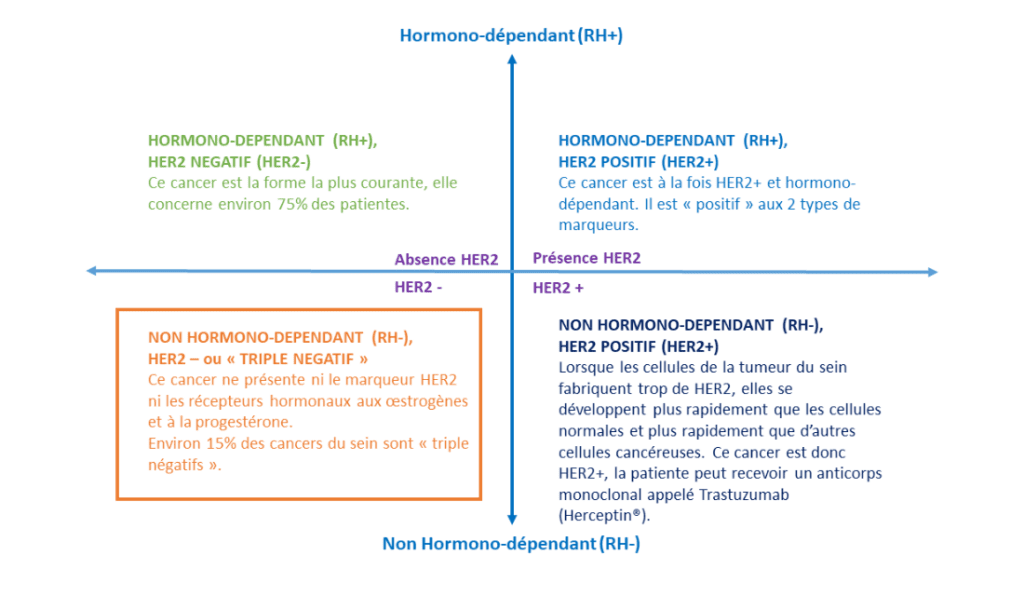
In January 2023, medical teams and researchers at the Centre Baclesse launched a new organoid clinical trial involving patients with triple-negative breast cancer. This type of breast cancer accounts for around 15% of the 61,000 breast cancers diagnosed in France each year(source INCa 2023), and is characterized by the absence of hormone receptors and the absence of overexpression of the HER2 receptor (a protein naturally present in the body). Highly aggressive, it mainly affects young women and carries a higher risk of recurrence.
The development of new methods for predicting response to treatment is a major challenge in triple-negative breast cancer today. Indeed, despite a good response to initial treatment, the rate of metastatic recurrence remains very high, with approximately 20-30% of relapses. Cancer can recur within the breast itself, in the form of local recurrence, or at a distance, in other organs or tissues, in the form of metastases.
Subtypes of breast cancer
Aim of the study
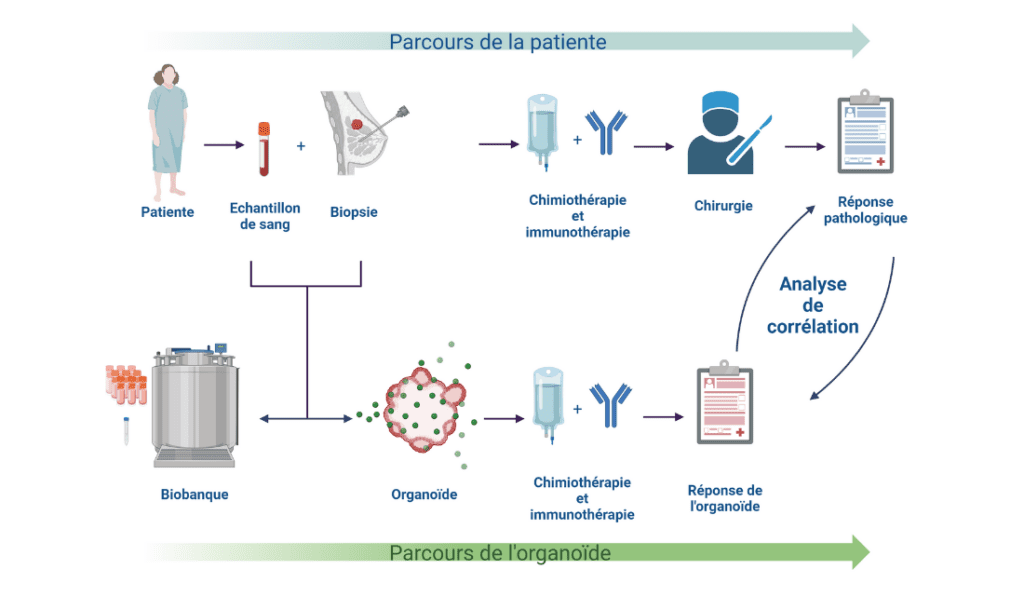
The aim of the TRIPLEX study is to "test different treatments on tumors taken from patients and reproduced in the laboratory, with the aim of determining which treatment will be most effective," explained Dr. George EMILE, medical oncologist and head of the Breast Cancer Unit at the Baclesse Center, at the launch of the study.
The study is divided into two stages:
- Step 1: Build up a collection of tumor and blood samples. These are processed by the ORGAPRED platform and the Tumorothèque de Caen Basse-Normandie, which also manages their storage.
- Step 2: Analyze the ex vivo response (tests performed outside the body in a laboratory) of tumor samples to treatments in order to develop functional tests and search for biomarkers predictive of treatment response.
The ORGAPRED platform is dedicated to the production of tumor organoids (also known as tumoroids) for predictive and research purposes. It is a platform of the University of Caen Normandie hosted by the François Baclesse Cancer Center within the Inserm Unit U1086 ANTICIPE. The scientific managers are : Laurent POULAIN and Louis-Bastien WEISWALD (Researchers).


ORGAPRED Laboratory
TRIPLEX study diagram
An update from Dr George Emile
- Several objectives have been pursued since the study began. Can you give us some initial details?
"The main objective was to determine whether we could develop an organoid solely from a biopsy; we estimated our success rate at 30%. To date, 37 patients have been included in the trial, and we have obtained organoids in 80% of cases, which is excellent news. However, they can only be kept in culture for a few weeks, and we are struggling to maintain their proliferation. We are therefore currently working on miniaturization processes so that we can evaluate the response to treatment as soon as the tumoroids appear. It is a real challenge to work with so little biological material. Indeed, the sample taken during a biopsy represents a very small fragment of tissue (the size of a needle), hence the challenge posed by the method used in this clinical protocol."
- What about your secondary objective, which was to determine whether the organoids developed correlated well with the original tumors?
"The characterization of organoids is underway but remains difficult with such a small amount of material. The team is continuing to refine its techniques for both the culture and characterization of organoids. This is a priority objective for 2024."
- What's next for the TRIPLEX trial?
"Treating organoids with the chemotherapy and immunotherapy drugs administered to patients. This crucial stage of the clinical trial will begin shortly, with the team currently finalizing the last steps to standardize the process. The team will then be able to focus on resistant organoids and test new treatments or therapeutic strategies (new treatment combinations) that can then be administered to patients. Ultimately, the hope is to implement personalized medicine for each patient and thus avoid treatments that are known to be ineffective. The cancer care of tomorrow is moving towards precision medicine, which requires not only the availability of effective alternative treatments, but also the identification of biomarkers that enable the selection of patients likely to benefit from these new therapeutic strategies.
Inclusion of patients in the clinical trial will extend over 4 years, and will involve a total of 163 patients.